Drug allergy to Anti Glaucoma medications
Introduction:
Ocular surface disorder (OSD) occur due to chronic, long term use of antiglaucoma medications (AGM). Instillation of topical AGMs for a period of three or more months has been found to cause significant subclinical inflammation with increased expression of HLA-DR on conjunctival epithelial cells[1] and secretion of pro-inflammatory cytokines by conjunctival cells.[2] Topical medication related OSD result in poorer compliance to treatment, compromised surgical results and also decreases the quality of life in glaucoma patients.[3] We present to you a patient of primary open angle glaucoma who developed severe drug allergy to brimonidine 0.2% eye drops after instilling it for 4 months.
Case Report:
A male, 63 years of age, known case of primary open angle glaucoma, presented to us in March 2020 with redness, itching, foreign body sensation and watering from both eyes for last 5 days which had increased gradually. Visual acuity in both eyes (BE) was 6/6. He had presence of lid edema and skin excoriation in BE (fig 2). He had significant congestion of conjunctiva and rest of the anterior segment findings were normal. IOP on applanation tonometer (AT) in Right eye (RE) was 15 mmHg and in Left eye (LE) was 13 mmHg. On fundus examination, RE showed vertical C:D ratio of 0.7 with inferior rim thinning and LE showed vertical C:D ratio of 0.5 with a healthy neuroretinal rim (Fig 1). It was our impression that the patient had developed allergy to topical AGM . He was advised to stop brimonidine 0.2 %, and was started on carboxymethyl cellulose 0.5% eye drops in both eyes for 1 month, periorbital application of lactocalamine lotion and tab levocitrizine-montelukast once daily for two weeks. Patient was reviewed after 2 weeks and was symptomatically doing much better. The lid edema and congestion had reduced significantly. At 1 month follow up all signs of allergy had resolved and patient was comfortable (fig3). IOP in RE on AT was 20 mmhg and in LE was 18 mmHg. Patient was started on preservative free brimonidine 0.2% and timolol 0.5 % eye drop and was asked to review after 3 months.
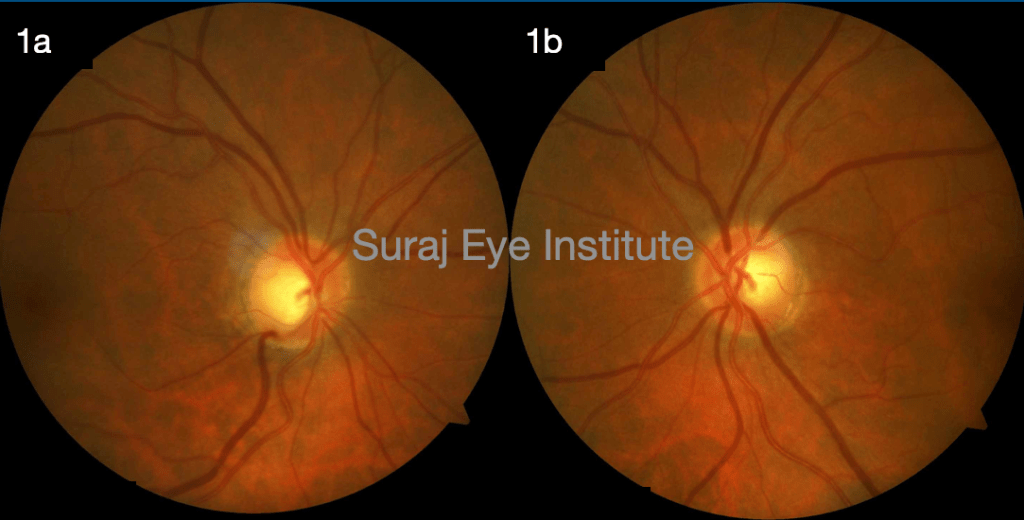
Fig 1b Left eye fundus photograph showing vertical C:D ratio of 0.5 with healthy NRR.


Discussion:
The prevalence of OSD has been reported in 59% of glaucoma cases with higher prevalence in patients using Benzalkonium chloride containing AGM.[4] Drug allergy is one of the important causes for non-compliance to treatment. Our patient developed severe drug allergy to brimonidine 0.2% eye drop after 4 months of use. It has been reported that the allergic reaction which occurs in 4.8–9% of patients on brimonidine eye drops is usually seen within the first 9 months. [5] We stopped AGM, for allergy to subside and started him on lubricating eye drop and anti-histaminic tablet systemically. IOP was monitored. Allergy to AGM should be promptly recognised and treated. Due to bothersome cosmetic adverse reactions, some patients may choose to discontinue glaucoma medications on their own. This can lead to reduced compliance and hence we need to warn the patients regarding possible side effects. Regular review has also to be explained so as to increase the compliance of the drug.
References:
- Arici MK, Arici DS, Topalkara A, Guler C. Adverse effects of topical antiglaucoma drugs on the ocular surface. Clin Exp Ophthalmol 2000;28(2):113-117. https://doi.org/10.1046/j.1442-9071.2000.00237.
- L, Montange T, Vejux A, Baudouin C, Bron AM, CreuzotGarcher C, Lizard G. Measurement of inflammatory cytokines by multicytokine assay in tears of patients with glaucoma topically treated with chronic drugs. Br J Ophthalmol 2007;91(1):29-32.http://dx.doi.org/10.1136/bjo.2006.101485
- PJ, Pouliquen P, Baudouin C. Prevalence of ocular symptoms and signs with preserved and preservative-free glaucoma medication. Br J Ophthalmol 2002;86(4):418-423.http://dx.doi.org/10.1136/bjo.86.4.418
- Leung EW, Medeiros FA, Weinreb RN. Prevalence of ocular surface disease in glaucoma patients. Journal of glaucoma. 2008 Aug 1;17(5):350-5.https://journals.lww.com/glaucomajournal/Abstract/2008/08000/Prevalence_of_Ocular_Surface_Disease_in_Glaucoma.3.aspx
- LeBlanc RP, for the Brimonidine Study Group 2. Twelvemonth results of an ongoing randomized trial comparing brimonidine tartrate 0.2% and timolol 0.5% given twice daily in patients with glaucoma or ocular hypertension. Ophthalmology 1998; 105: 1960–1967.https://doi.org/10.1016/S0161-6420(98)91048-X
